Tirzepatide vs. GLP-1 Agonists

Tirzepatide vs. GLP-1 Agonists (Semaglutide)
Over the past decade, the landscape of metabolic health, type 2 diabetes management, and clinical weight loss has undergone a seismic shift. Moving away from therapies that solely manipulate insulin or rely entirely on central nervous system stimulants, modern medicine has turned its focus to the gut-brain axis. At the forefront of this revolution are incretin mimetics—medications that mimic the body’s natural metabolic hormones.
Currently, two primary compounds dominate this space: Semaglutide (the most widely recognized GLP-1 receptor agonist) and Tirzepatide (a novel dual-agonist). While both have shown unprecedented efficacy in clinical settings, they operate through different biological pathways and yield slightly different outcomes.
This comprehensive guide explores the physiological mechanisms, clinical efficacy, side effect profiles, and distinct differences between Tirzepatide and the broader GLP-1 classification, specifically Semaglutide.

Part 1: The Biological Mechanisms
To understand how these compounds differ, it is essential to first understand the natural incretin hormones they are designed to replicate. Incretins are hormones secreted by the gut in minutes after eating. They play a crucial role in managing how the body utilizes and stores energy.
The Foundation: How GLP-1 Works (Semaglutide)
Semaglutide belongs to the GLP-1 (Glucagon-Like Peptide-1) receptor agonist class. When a person eats, the intestines naturally release GLP-1. Semaglutide is a synthetic version of this hormone, engineered with a structural modification that prevents it from being rapidly broken down by enzymes in the body, allowing it to remain active for a full week.
GLP-1 impacts the body through three primary mechanisms:
-
Pancreatic Action: It stimulates the beta cells in the pancreas to release insulin in a strictly glucose-dependent manner. This means it only prompts insulin release when blood sugar levels are elevated, drastically reducing the risk of hypoglycemia (dangerously low blood sugar). Simultaneously, it suppresses the release of glucagon, the hormone that prompts the liver to release stored sugar.
-
Gastric Emptying: GLP-1 significantly slows down the rate at which the stomach empties its contents into the small intestine. This delayed gastric emptying leads to a prolonged sensation of physical fullness after meals, naturally reducing caloric intake.
-
Neurological Appetite Regulation: Perhaps most significantly for weight management, GLP-1 receptors are highly concentrated in the hypothalamus—the region of the brain that regulates hunger and satiety. The medication directly binds to these receptors, dampening appetite signals and reducing “food noise” or cravings.
The Evolution: The Twin-Incretin Effect (Tirzepatide)
Tirzepatide represents the next generation of metabolic therapy. It is a dual GIP/GLP-1 receptor agonist. This means that in addition to mimicking GLP-1, it also mimics a second, highly potent incretin hormone called GIP (Glucose-Dependent Insulinotropic Polypeptide).
For years, GIP was largely ignored in obesity research because it appeared to lose its insulin-stimulating efficacy in patients with type 2 diabetes. However, researchers discovered that when GIP is combined with GLP-1, it acts synergistically, creating a compounding effect that is greater than the sum of its parts.
The addition of GIP introduces several novel physiological benefits:
-
Enhanced Lipid Metabolism: GIP receptors are abundant in adipose (fat) tissue. Activation of these receptors improves the body’s ability to clear dietary lipids (fats) from the bloodstream, preventing ectopic fat deposition (fat stored in the liver or around organs).
-
White Adipose Tissue Regulation: GIP appears to increase insulin sensitivity directly within white fat cells, improving metabolic markers independent of weight loss.
-
Tolerability Buffering: Interestingly, GIP signaling in the central nervous system has been shown to potentially buffer against the nausea commonly associated with GLP-1 receptor activation. This allows Tirzepatide to be titrated to highly effective doses while maintaining a manageable side effect profile.
Part 2: Head-to-Head Clinical Efficacy
While physiological theory is important, the true test of these compounds lies in rigorous, large-scale clinical trials. Both drugs have been extensively studied under massive clinical programs: the STEP program for Semaglutide and the SURPASS / SURMOUNT programs for Tirzepatide.
Efficacy in Weight Management
Both medications have redefined the benchmarks for non-surgical weight loss, producing results that approach those of bariatric surgery.
-
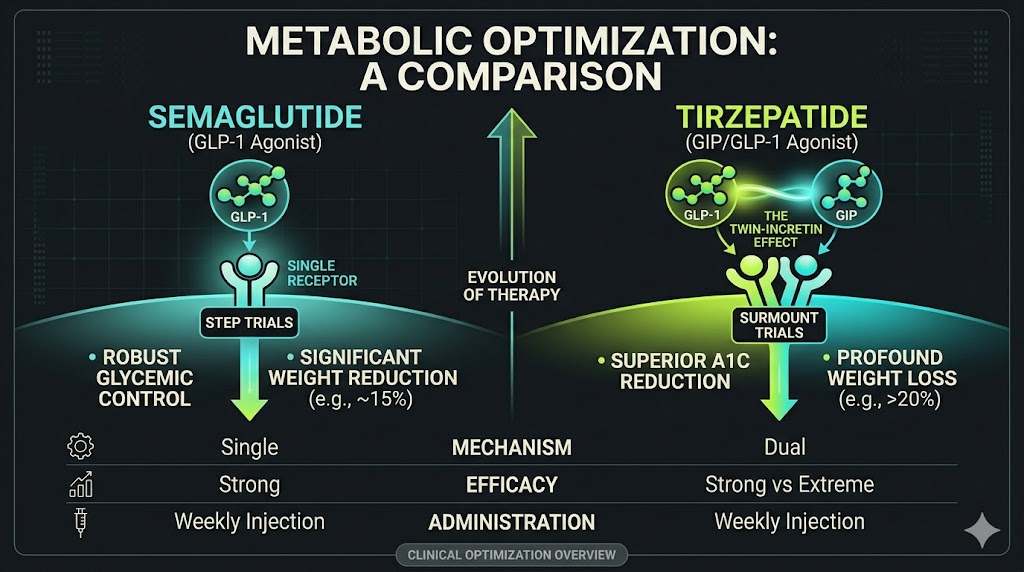
Semaglutide (GLP-1): In the landmark STEP 1 trial, patients without type 2 diabetes taking the maximum dose (2.4 mg weekly) alongside lifestyle interventions lost an average of 14.9% of their total body weight over 68 weeks. It established the GLP-1 class as the gold standard for clinical obesity management.
-
Tirzepatide: In the SURMOUNT-1 trial, the dual-agonist approach demonstrated even more profound results. Patients on the highest dose (15 mg weekly) achieved an unprecedented average weight loss of 20.9% to 22.5% over 72 weeks. Furthermore, a significantly higher percentage of participants on Tirzepatide achieved weight loss targets of 15%, 20%, and 25% compared to historical GLP-1 data.
The Verdict on Weight Loss: While Semaglutide provides exceptional, life-altering weight reduction for many, clinical data consistently indicates that Tirzepatide yields a higher absolute percentage of total body weight lost, largely attributed to the synergistic addition of the GIP receptor activation.
Efficacy in Glycemic Control (A1C Reduction)
For patients managing type 2 diabetes, the primary metric of success is the reduction of Hemoglobin A1C (a measure of average blood sugar over three months).
-
Semaglutide (GLP-1): Known for robust glycemic control, the 1.0 mg and 2.0 mg doses typically reduce A1C by roughly 1.5% to 1.8%, effectively bringing many patients back into a non-diabetic or pre-diabetic range.
-
Tirzepatide: The SURPASS trials pitted Tirzepatide directly against 1.0 mg Semaglutide. Tirzepatide demonstrated superiority across all doses, with the highest dose reducing A1C by up to 2.3%. Notably, over 50% of participants on the highest dose of Tirzepatide achieved normoglycemia (an A1C of under 5.7%).
Part 3: Cardiovascular and Secondary Health Benefits
Obesity and type 2 diabetes are rarely isolated conditions; they are deeply intertwined with cardiovascular disease, renal (kidney) function, and other metabolic syndromes. Both medications have proven to be far more than just “weight loss drugs.”
Cardiovascular Outcomes
The broader GLP-1 classification has a massive head start in cardiovascular data. Massive, multi-year cardiovascular outcome trials (CVOTs) have definitively proven that Semaglutide reduces the risk of major adverse cardiovascular events (MACE), such as heart attack and stroke, by roughly 20% in high-risk patients. This has revolutionized cardiac care, prompting cardiologists to prescribe GLP-1s independent of weight loss goals.
Tirzepatide’s cardiovascular outcome trials (such as the SURPASS-CVOT) are ongoing. While early surrogate markers (like reductions in blood pressure, visceral fat, and systemic inflammation) are highly promising, Tirzepatide does not yet have the decade of definitive, peer-reviewed cardiovascular safety data that the GLP-1 class possesses.
Emerging Secondary Benefits
-
Sleep Apnea: Recent data for Tirzepatide shows it can effectively resolve moderate-to-severe obstructive sleep apnea in up to 50% of patients, largely due to significant reductions in airway fat deposits.
-
Liver Health: Both compounds are showing tremendous promise in treating MASH (Metabolic dysfunction-associated steatohepatitis) by reducing liver fat and halting fibrosis progression.
Part 4: Side Effect Profiles and Tolerability
Because both medications manipulate gastric motility and central nervous system signaling, their side effect profiles are remarkably similar, primarily affecting the gastrointestinal (GI) tract.
Common Adverse Events
-
Nausea: The most frequently reported side effect for both drugs, typically occurring during the first few days after an injection or when stepping up to a higher dose.
-
Vomiting and Diarrhea: Less common than nausea but frequently reported.
-
Constipation: A direct result of delayed gastric emptying and reduced fluid intake.
-
Fatigue: Often secondary to a drastic reduction in caloric intake.
Differences in Tolerability
Despite Tirzepatide resulting in greater absolute weight loss, its side effect profile is generally comparable to Semaglutide. The theorized reason is that GIP activation helps mitigate the nausea typically induced by intense GLP-1 activation. Consequently, many clinicians note that patients who experience severe, unmanageable nausea on Semaglutide may occasionally tolerate Tirzepatide better, though this is highly individual.
Sarcopenia (Muscle Loss)
A significant consideration with both medications is the composition of the weight lost. Because these drugs cause rapid, profound weight reduction, a portion of that loss will inevitably be lean muscle mass (sarcopenia). Without concurrent resistance training and adequate protein intake, patients risk lowering their basal metabolic rate, which can complicate long-term weight maintenance if the medication is ever discontinued.
Part 5: Dosing, Administration, and Titration
Both medications are administered via a once-weekly subcutaneous injection (typically into the abdomen, thigh, or upper arm) using a pre-filled pen device.
The most critical aspect of administration for both compounds is titration.
Because these medications drastically alter gastric emptying, starting at a high dose will almost certainly result in severe, debilitating gastrointestinal distress. Patients must begin at a “loading dose”—a non-therapeutic dose designed simply to acclimate the body to the compound.
-
GLP-1 Titration: Typically increases every 4 weeks.
-
Tirzepatide Titration: Also increases in 4-week intervals, allowing the body to adjust to both the GLP-1 and GIP receptor activation.
Patience during the titration phase is paramount. Many patients do not experience significant appetite suppression or weight loss until they reach the middle or upper tiers of the dosing schedule, which can take two to three months.
Conclusion: Navigating the Future of Metabolic Therapy
The advent of both standard GLP-1 agonists and dual-incretin mimetics represents a paradigm shift in how modern medicine approaches metabolic dysfunction.
Semaglutide stands as the proven, foundational titan of the GLP-1 class. It boasts a massive wealth of long-term cardiovascular outcome data, widespread clinical familiarity, and a proven track record of life-changing efficacy for both diabetes management and clinical obesity.
Tirzepatide represents the cutting-edge evolution of this science. By harnessing the twin-incretin effect of both GLP-1 and GIP, it has consistently demonstrated superior reductions in both total body weight and A1C in head-to-head clinical trials, pushing the boundaries of what pharmacological intervention can achieve.
Ultimately, the choice between these compounds is a highly individualized clinical decision. It must be made under the direct, ongoing supervision of a qualified healthcare provider who can evaluate a patient’s complete metabolic profile, cardiovascular risk factors, and personal tolerance to navigate this powerful new era of metabolic medicine.